Examination of novel cAMP signaling pathways in inflammatory diseases of the skin
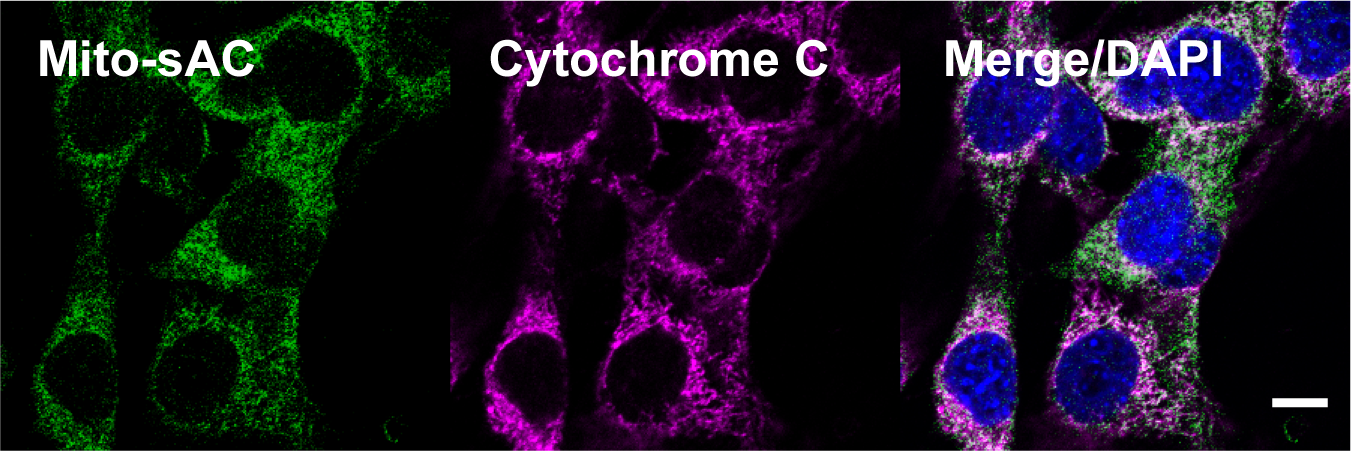
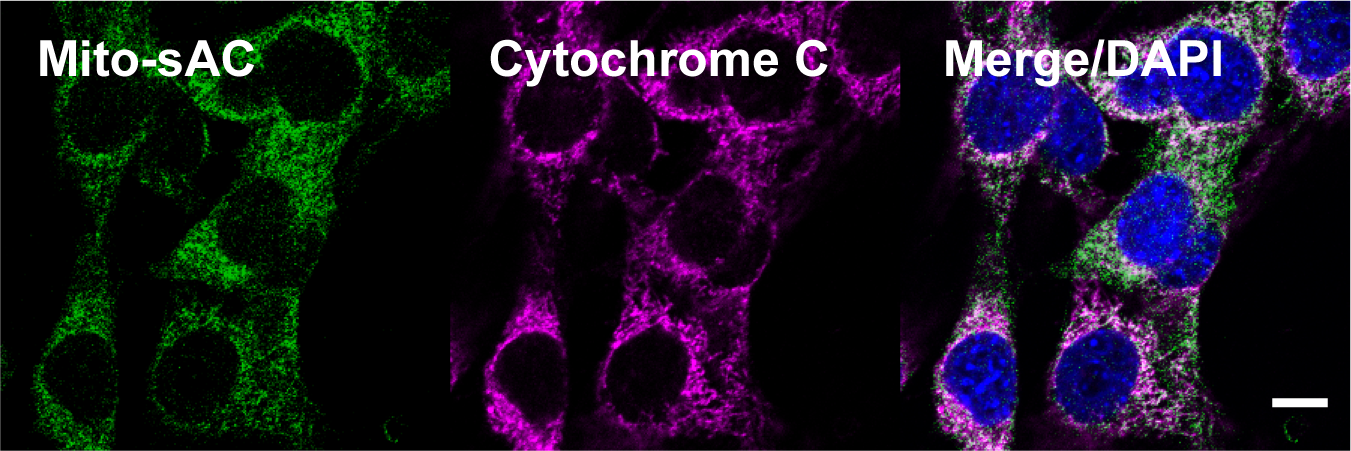
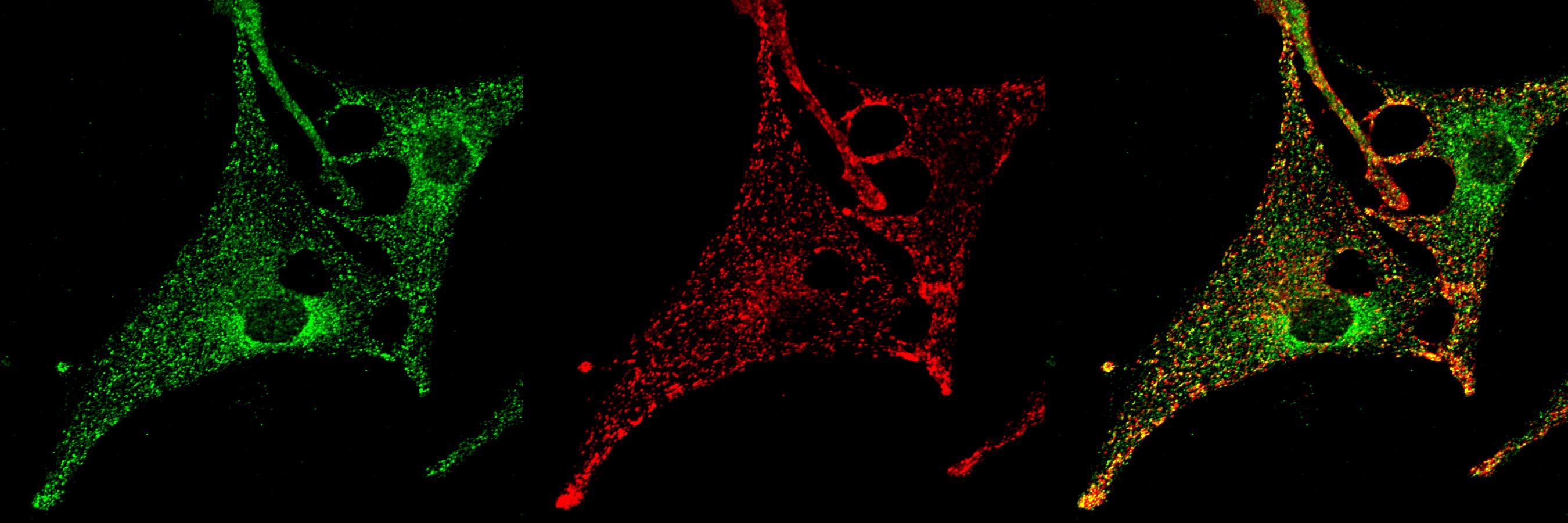
cAMP has distinct effects in multiple inflammatory cells and drugs that target cAMP signaling have shown promise as therapeutics for diseases such as psoriasis and atopic dermatitis. However, the exact mechanisms by which cAMP influences inflammatory cells in the skin remains poorly understood. We have taken a multipronged approach to dissect the role of cAMP in different inflammatory cells in the skin. We have established that specific sources of cAMP play distinct roles in the activation of T helper cells during psoriasis, atopic dermatitis, and allergic contact dermatitis. We are currently working on establishing the mechanisms by which these distinct cAMP signaling cascades regulate T helper cell development and activation. We are also leveraging our association with the Tri-Institutional Therapeutics Discovery Institute to develop a class of non-steroidal topical therapeutics for the treatment of inflammatory skin diseases.